Orthopedic Product Development at Exactech
Led surgeon-driven instrument and implant development across CAD, DFM, testing, manufacturing, and 510(k) support through 7 concurrent projects.
Role
Product Design Engineer
Industry
Healthcare
Duration
6 Months

Overview
During my time at Exactech, I joined as a Product Design Engineering Intern on the Upper Extremities team and continued on as a part-time Product Design Engineer, supporting surgeon-driven development efforts across instruments, implants, testing, manufacturing readiness, and regulatory documentation. Across 7 concurrent projects, I worked at the intersection of clinical input, CAD, manufacturability, validation, and documentation within a regulated orthopedic medical device environment.
Note: All visuals, models, and descriptions shown are sanitized and redacted to respect company confidentiality and intellectual property.
Surgeon-specific Special Instruments development (concept to market release)
Inlay / Onlay humeral stem interference anatomy study (Siemens NX analysis)
Shoulder Testing History Matrix creation for fatigue testing standardization
Lifecycle and durability test method development
Manufacturing print updates and tolerance stack-ups
Process Flow Chart (PFC) remediation for suppliers and internal manufacturing
510(k) support through validation labs and clinical documentation
Special Instruments: Surgeon-Driven Products
At Exactech, Special Instruments represent a differentiated approach to surgeon collaboration, where standard-line orthopedic instruments are selectively customized to match individual surgeon ergonomics, preferences, and operating workflows. Rather than designing entirely new systems, these instruments adapt proven platforms to clinical nuance, enabling personalization while maintaining manufacturability, regulatory alignment, and compatibility with existing trays.
I managed 7 Special Instruments from concept through final delivery, serving as the coordination point between consulting surgeons, design engineering, manufacturing, and sales. This work spanned early requirement capture, design input definition, iterative CAD development in Siemens NX, manufacturability alignment with the Development Shop, quote coordination, and delivery planning. Each instrument balanced surgeon-specific needs with design-for-manufacturing, safety, and standardization constraints, providing a rare opportunity to execute customized medical device development at scale.
Case Study: Ergo Drill Guide Redesign
Challenge: One representative project was the redesign of an Ergo Drill Guide, where a consulting surgeon requested a lightweight drill guide compatible with a 3.2 mm K-wire while maintaining alignment accuracy and safe handling during surgery. The existing design presented overlapping holes that introduced manufacturability challenges and raised safety concerns.
Solution: To address this, I worked with design engineers to iterate the CAD by enlarging the K-wire feature, adding defined female “toast” geometries for tool engagement, and reducing overall body thickness to improve ergonomics without compromising strength. These changes resolved the DFM and safety issues while meeting the surgeon’s clinical requirements, demonstrating how targeted geometric adjustments can materially improve both usability and manufacturability.

Shoulder Implant Research & Design Standardization
I contributed to research initiatives focused on improving how shoulder implants are evaluated, analyzed, and designed across Exactech’s portfolio.
One major initiative was the Inlay / Onlay Stem Interference Anatomy Study, where I evaluated how different humeral stem geometries interacted with patient anatomy. Using Siemens NX, I analyzed proximal and distal interference across multiple bone models, visualizing how implant geometry engaged cortical bone under different configurations. This analysis helped translate simulated anatomical data into actionable design insight, informing dimensional refinements and manufacturability decisions rather than relying solely on anecdotal clinical feedback.
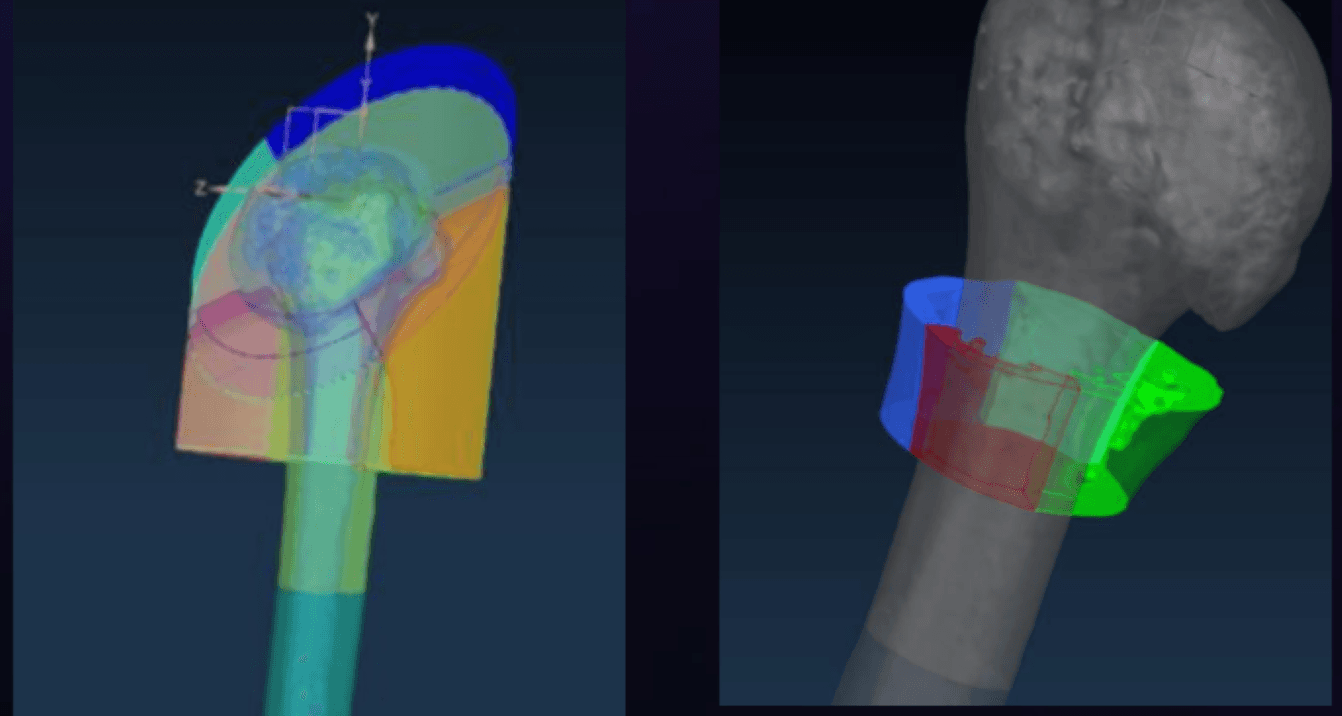
Clinical-Informed Durability Testing
I led durability and lifecycle testing efforts for a Special Extended Klimo Inserter, a surgeon-requested variant designed to support specific impaction and assembly workflows during shoulder arthroplasty. This work sat at the intersection of clinical technique, mechanical loading, and regulatory test requirements.
During durability testing, the instrument failed under impaction forces reaching 1,127 lbf, revealing a gap between standard test assumptions and real intraoperative use. In response, I authored and executed a custom assembly/disassembly lifecycle test protocol that more accurately replicated the sequence, grip, and loading conditions described in the operative technique.
By reframing the test method around how surgeons actually use the instrument, rather than idealized lab conditions, we were able to generate more clinically relevant data to guide design decisions and downstream validation. This effort reinforced the importance of embedding clinical context directly into verification and validation strategies for surgical instruments.

Manufacturing Readiness & Print Updates
To support production scalability and reduce downstream nonconformances, I worked closely with Manufacturing Engineering and Quality to resolve tolerance-driven issues in existing instrument lines. This work focused on translating design intent into manufacturable, inspection-friendly drawings while ensuring compliance with existing verification and validation frameworks.
Case Study: Validation Continuity for Modified Tray Trials
For updated Ergo Tray Trials, I compiled FORM-000130 to demonstrate that existing V&V testing remained applicable despite the design modification. This ensured regulatory continuity while avoiding redundant testing, accelerating release timelines and maintaining compliance.
Case Study: Print Update for Steinmann Pins
I led a tolerance stack-up analysis on the Steinmann pin assembly to identify the root cause of recurring nonconformances. By widening the tolerance band through dimensional reallocation, I eliminated a recurring Use-As-Is disposition and reduced inspection rejections without compromising functional performance.
In manufacturing, the most robust designs are often the simplest—clear tolerances, fewer failure modes, and alignment between design intent and production reality.
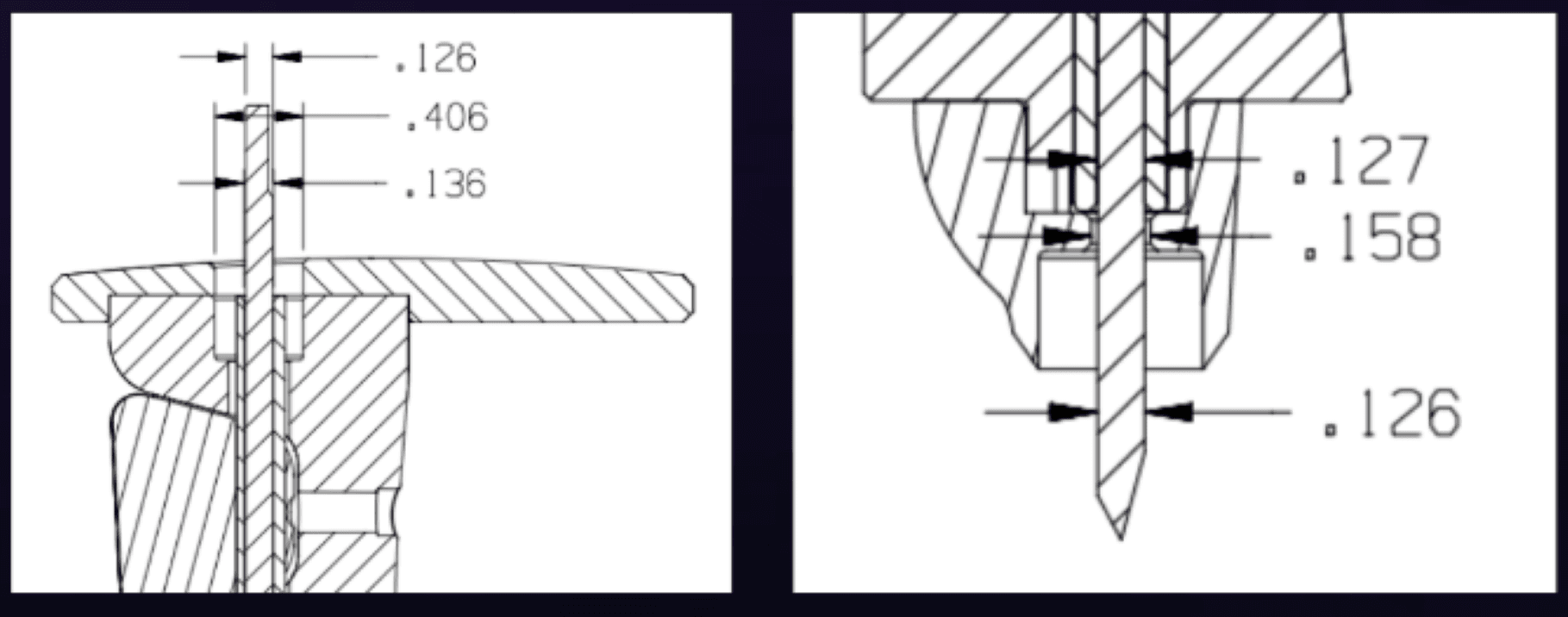
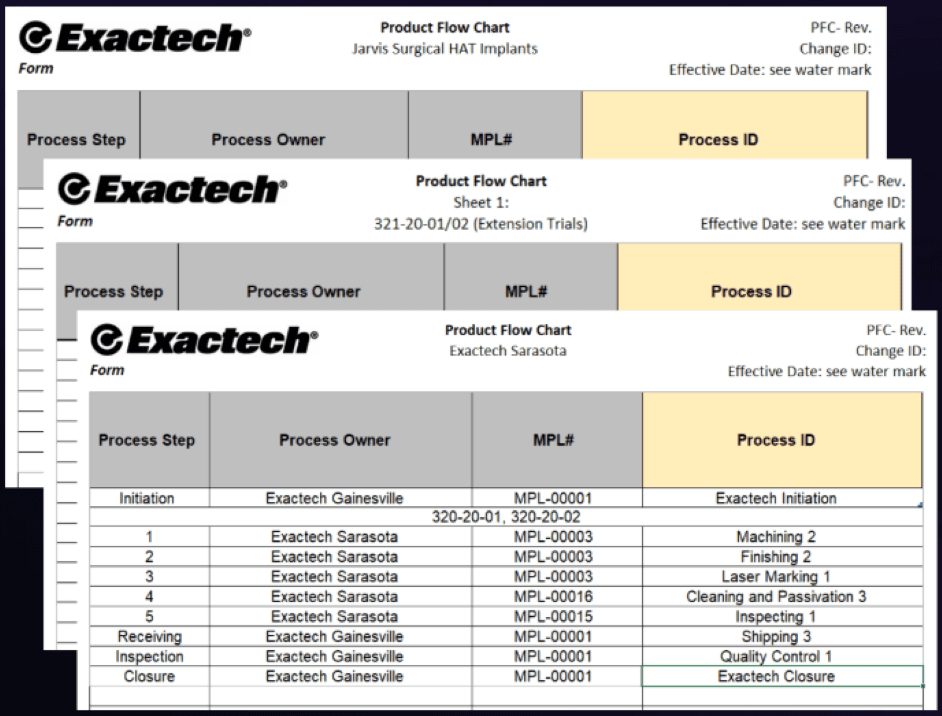
Supply Chain & Process Documentation
To support manufacturing continuity and regulatory readiness, I owned updates to process documentation across multiple implant and instrument product lines. This work focused on reconciling how products were actually being built across vendors with how those processes were documented internally—closing gaps that could introduce risk, delays, or audit findings.
I remediated and updated Process Flow Charts for HAT implants, surgical instruments, screws, and Ergo Tray Trials, ensuring each reflected the correct sequence of manufacturing, inspection, and finishing steps across internal and external suppliers.
I cross-checked PFCs against Manufacturing Process Lists (MPLs) and vendor-provided documentation to resolve discrepancies and ensure traceability. This required close collaboration with Exactech Sarasota and Gainesville, as well as external suppliers including Jarvis Surgical, Sheffield, InTech Medical, and AutoCam.
End User Feedback & Clinical Evaluation
To ensure surgeon and clinician input directly informed product refinement and regulatory submissions, I played a key role in capturing, synthesizing, and presenting end-user feedback across live surgical and validation environments.
Clinical Case Documentation & Presentation: I compiled and structured case presentations on bone loss and disasterplasty for Exactech’s Annual Clinical Evaluators’ Meeting, translating intraoperative observations into clear, reviewable technical narratives for cross-functional teams.
Validation Lab Feedback Capture: I assisted with documentation and minute-taking during a Glenoid Reconstruction Prosthesis validation lab, observing live use of instruments and guides in simulated surgical conditions. I translated surgeon commentary—covering fit, tactile feedback, drill guidance, and depth control—into structured feedback suitable for downstream design evaluation.
Regulatory-Ready Documentation: All captured surgeon feedback was formalized for inclusion in 510(k) submission materials, ensuring traceability between clinical observations, design decisions, and regulatory evidence.

Other projects

Arco: Transformable Desk for Small-Space Living
Arco is a configurable desk system designed for students living in small spaces, created to help people physically and mentally transition between modes throughout the day.

Autonomous Car
Designing and building an autonomous mobile robot with real-time control, localization, and sensing, from system architecture to competition-ready deployment.

Biofluid Analyzer
Developing a compact bedside urine output monitoring system for critical care, from clinician pain point to deployed prototype and formal pilot study.

Product Growth at PrimeEventZ
Leading product definition and real-world deployment of PrimeEventZ, an AI-powered platform unifying event operations from planning through execution at any scale.